The 2026 Regulatory Shakeup is transforming U.S. medical device compliance. Learn how FDA’s new Quality Management System Regulation (QMSR) impacts manufacturers, distributors, and suppliers—and how to prepare.

Understanding the 2026 Regulatory Shakeup in U.S. Medical Device Oversight
The 2026 Regulatory Shakeup marks one of the most significant changes to U.S. medical device regulation in decades. Beginning February 2, 2026, the U.S. Food and Drug Administration will fully enforce the Quality Management System Regulation (QMSR), fundamentally changing how medical device compliance is evaluated in the United States.
For manufacturers, distributors, and suppliers of medical supplies, this is more than a regulatory update—it is a structural shift. The FDA is moving away from its long-standing Quality System Regulation (QSR) and aligning U.S. requirements with ISO 13485, the globally recognized standard for medical device quality management systems.
This change reflects the reality of today’s medical device industry: global supply chains, outsourced manufacturing, software-driven devices, and increasing patient safety expectations. Companies that treat the 2026 Regulatory Shakeup as a box-checking exercise risk compliance failures, warning letters, and lost market access. Those that prepare early can reduce risk, streamline operations, and strengthen trust with regulators and customers alike.
Why FDA Introduced the Quality Management System Regulation (QMSR)
The FDA introduced QMSR to modernize and harmonize U.S. medical device regulation. For years, manufacturers operating globally have had to maintain two parallel quality systems—one for FDA QSR and another for ISO 13485. This duplication created inefficiencies, confusion, and unnecessary compliance costs.
Under the 2026 Regulatory Shakeup, FDA aims to:
- Align U.S. requirements with international standards
- Improve consistency in inspections and enforcement
- Enhance patient safety through modern risk management principles
- Reduce regulatory burden without lowering expectations
By adopting ISO 13485 by reference, FDA brings U.S. oversight in line with regulators in Europe, Canada, Japan, and other major markets.
Key Compliance Dates and Enforcement Timeline
Understanding the timeline is critical for compliance planning:
| DateMilestone | |
| January 31, 2024 | Final QMSR rule published |
| February 2, 2026 | QMSR becomes fully enforceable |
| 2024–2026 | Transition period for manufacturers |
| Post-2026 | FDA inspections conducted under QMSR framework |
While enforcement begins in 2026, FDA expects companies to demonstrate active transition efforts well before that date.
What Is the FDA Quality Management System Regulation (QMSR)?
The Quality Management System Regulation replaces most of 21 CFR Part 820 with a new framework that incorporates ISO 13485:2016 by reference. Rather than prescribing highly detailed requirements, QMSR emphasizes a system-based, risk-driven approach to quality.
From QSR to QMSR: What Changed and Why It Matters
Under QSR, compliance focused heavily on procedural adherence and documentation checklists. QMSR shifts the focus to:
- Risk-based decision-making
- Lifecycle management of devices
- Integration of suppliers and outsourced processes
- Continuous improvement
This shift means FDA will assess not just whether you have procedures, but how effectively your quality system works in practice.

Alignment With ISO 13485: A Strategic Shift
ISO 13485 is already familiar to many U.S. companies that sell internationally. By aligning with this standard, the 2026 Regulatory Shakeup:
- Reduces duplication of audits
- Simplifies global compliance strategies
- Improves clarity for multinational suppliers
However, alignment does not mean FDA oversight becomes easier. FDA will still enforce U.S.-specific requirements, including complaint handling, labeling controls, and post-market surveillance.
Who Is Affected by the 2026 Regulatory Shakeup?
Medical Device Manufacturers
Manufacturers bear primary responsibility under QMSR. This includes:
- Design and development controls
- Risk management throughout the product lifecycle
- Corrective and preventive actions (CAPA)
- Post-market monitoring
Companies that have relied on minimal QSR compliance will face the steepest learning curve.
Distributors, Importers, and Suppliers
The 2026 Regulatory Shakeup expands expectations across the supply chain. Distributors and suppliers must demonstrate:
- Robust supplier qualification processes
- Traceability of components and materials
- Effective communication with manufacturers
FDA inspections may increasingly examine supplier controls as part of manufacturer audits.
Contract Manufacturers and Service Providers
Outsourced manufacturing, sterilization, testing, and software services are now squarely in scope. Under QMSR, manufacturers must ensure external providers operate within the same quality framework.
Core Requirements Under FDA’s QMSR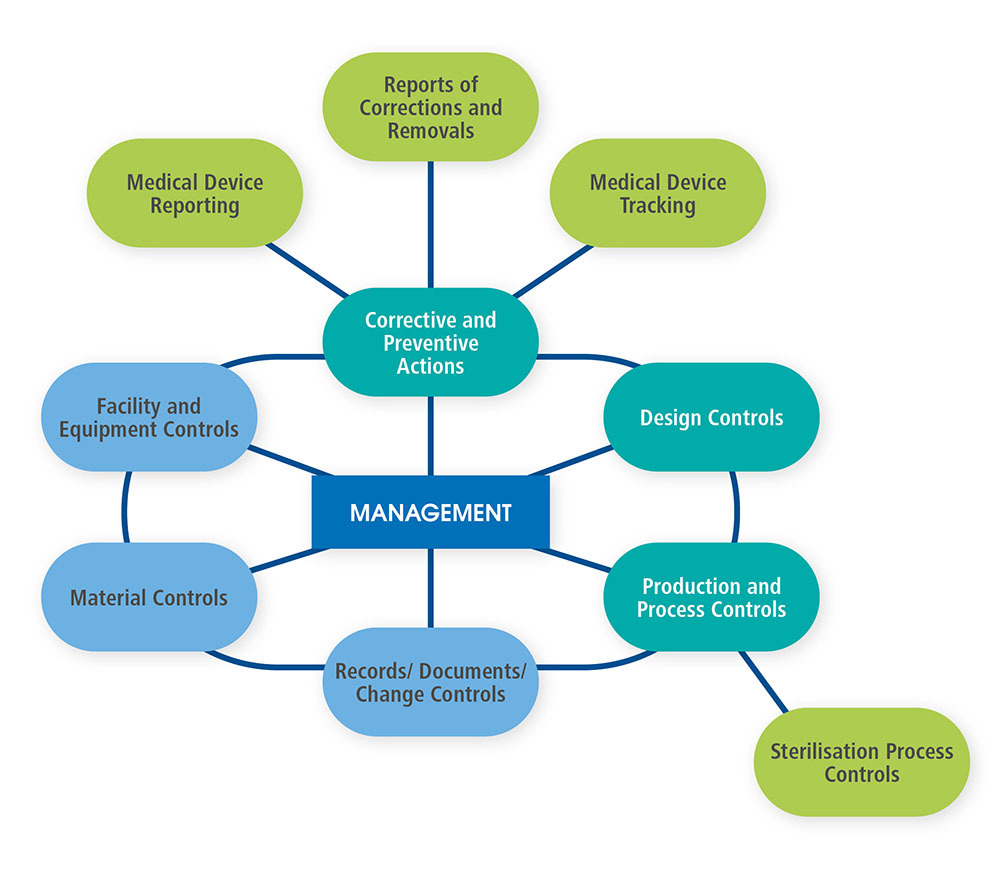
Key FDA Policies form Medical Device Manufacturer Inspections | NSF
Risk Management and Design Controls
Risk management is no longer a standalone activity. Under QMSR, it must be integrated into:
- Design and development
- Supplier selection
- Production and process controls
- Post-market surveillance
FDA expects risk management to be documented, justified, and continuously updated.
Documentation, Recordkeeping, and Traceability
While QMSR reduces some prescriptive documentation, it raises expectations for clarity and consistency. Records must demonstrate:
- Design intent and changes
- Traceability from requirements to verification
- Clear decision-making rationale
Poor documentation remains one of the most common FDA inspection findings.
Supplier Controls and Purchasing Management
Supplier oversight is a major focus of the 2026 Regulatory Shakeup. Companies must show:
- Risk-based supplier qualification
- Ongoing performance monitoring
- Clear quality agreements
Failure to control suppliers can result in findings—even if the issue occurred outside your facility.
How QMSR Redefines FDA Inspections and Audits
What Investigators Will Focus on in 2026
FDA inspections under QMSR will be more holistic and systems-focused. Inspectors will evaluate:
- How risks are identified and mitigated
- Whether quality objectives align with business decisions
- Effectiveness of CAPA systems
- Integration of suppliers and contractors
Expect fewer checklist-style inspections and more probing questions about how your system actually works.
Compliance Challenges for U.S. Medical Device Companies
Common Gaps Between QSR and ISO 13485
Many U.S. companies assume they are “mostly compliant” because they meet QSR. Common gaps include:
- Weak risk management integration
- Informal supplier oversight
- Limited management review effectiveness
- Insufficient post-market data analysis
Closing these gaps requires both technical updates and cultural change.
Preparing Your Organization for the 2026 Regulatory Shakeup
Step-by-Step Transition Strategy
- Conduct a QMSR gap assessment
- Update quality manuals and procedures
- Integrate ISO 13485 risk principles
- Strengthen supplier management
- Perform internal audits under QMSR criteria
Training, Change Management, and Culture
Training is critical. Employees must understand why changes are happening, not just what is changing. Organizations that foster a quality-focused culture will adapt faster and perform better during inspections.
Business Benefits of Early QMSR Adoption
While compliance is mandatory, early adoption offers real advantages:
- Fewer audit redundancies
- Stronger supplier relationships
- Improved product quality and safety
- Increased confidence from customers and partners
In many cases, QMSR alignment reduces long-term operational costs.
FAQs on FDA QMSR and the 2026 Regulatory Shakeup
1. When does FDA QMSR enforcement begin?
Full enforcement begins February 2, 2026.
2. Does QMSR completely replace QSR?
Yes, most of 21 CFR Part 820 is replaced, with some U.S.-specific requirements retained.
3. Is ISO 13485 certification required?
Certification is not mandatory, but compliance with ISO 13485 requirements is.
4. Are distributors subject to FDA inspections under QMSR?
Indirectly, yes—especially through supplier and purchasing controls.
5. What happens if a company is not ready by 2026?
Companies risk FDA Form 483 observations, warning letters, or enforcement action.
6. Where can companies find official FDA guidance?
FDA publishes updates and guidance at https://www.fda.gov.
Conclusion: Turning Compliance Into Competitive Advantage
The 2026 Regulatory Shakeup is not just a regulatory hurdle—it is an opportunity. FDA’s Quality Management System Regulation pushes the U.S. medical device industry toward global alignment, stronger risk management, and better patient outcomes.
Manufacturers, distributors, and suppliers that act early will not only avoid compliance pitfalls but also position themselves as trusted, high-quality partners in an increasingly competitive market. In the end, QMSR is about more than rules—it is about building systems that work, protect patients, and support sustainable growth.