Understanding the FDA’s 2026 QMSR Shift
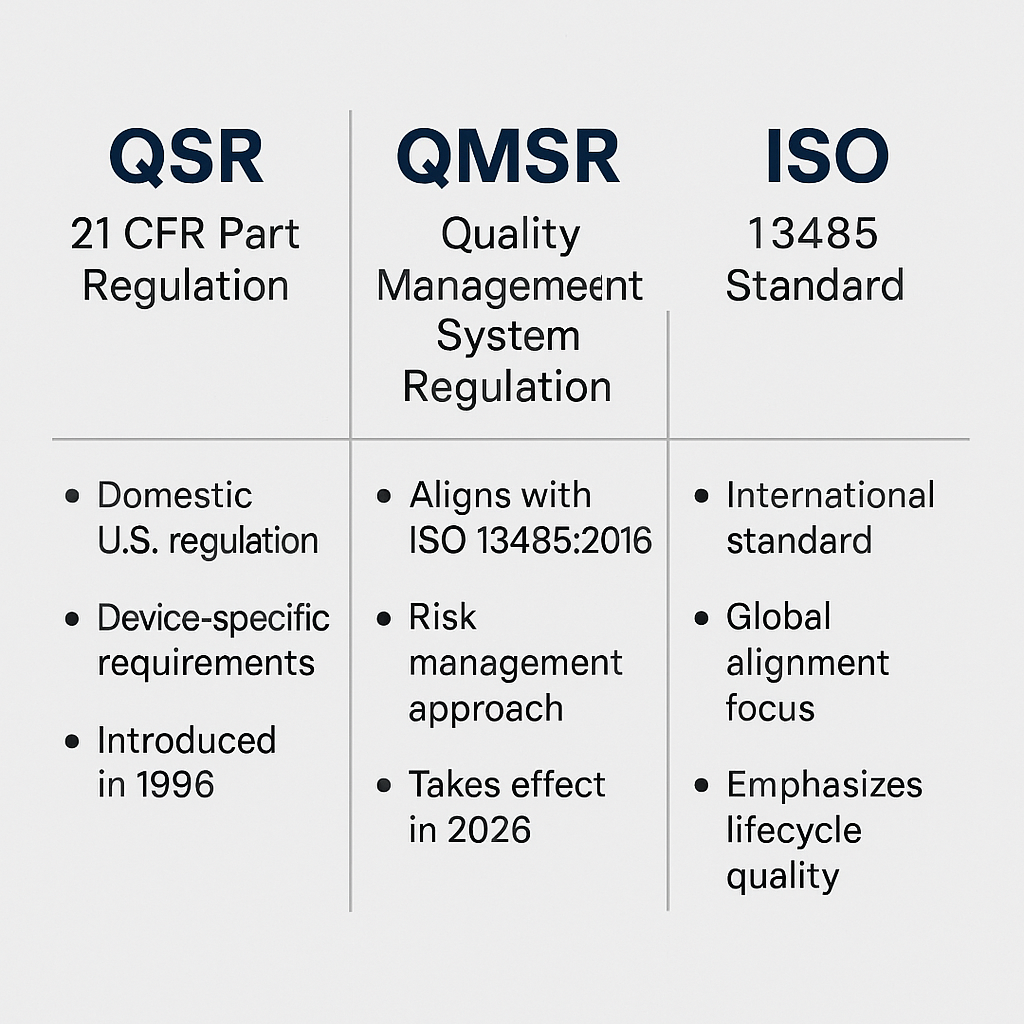
In January 2026, the U.S. Food and Drug Administration (FDA) will replace the long-standing Quality System Regulation (QSR, 21 CFR Part 820) with the Quality Management System Regulation (QMSR) — a landmark regulatory evolution designed to harmonize U.S. medical device quality standards with ISO 13485:2016.
This alignment represents the FDA’s vision of reducing redundant audits, improving international market access, and strengthening product safety through globally recognized quality practices. For decades, QSR served as the backbone of U.S. medical device manufacturing, but the rise of global trade and international standards has prompted the need for modernization.
What Is the QMSR (Quality Management System Regulation)?
The QMSR establishes a new framework where medical device manufacturers can operate under a globally unified quality management system, following ISO 13485 principles. The rule ensures product consistency, risk management, and post-market vigilance across every stage of the device lifecycle.
Under the QMSR, manufacturers must maintain robust quality documentation, implement risk-based decision-making, and align design, development, and production processes with international best practices.
Why Is the FDA Replacing the QSR (Quality System Regulation)?
The existing QSR, introduced in 1996, became increasingly misaligned with modern quality frameworks and global expectations. While it emphasized device safety, it lacked integration with the risk management and lifecycle approachemphasized by ISO 13485.
The 2026 QMSR aims to:
- Eliminate overlapping audits between the FDA and other regulators.
- Foster smoother international trade for U.S. device manufacturers.
- Enhance post-market surveillance and quality system transparency.
- Encourage proactive quality control and risk-based management.
Timeline and Implementation of the 2026 QMSR Rule
The final rule was published in early 2024, with an official compliance deadline in February 2026. Between now and then, manufacturers are expected to:
- Conduct internal audits comparing current QSR practices to ISO 13485.
- Update procedures, documentation, and training to meet QMSR requirements.
- Collaborate with notified bodies and the FDA for guidance and alignment.
The Link Between QMSR and ISO 13485:2016
Aligning U.S. Regulations with Global Standards
The QMSR fully integrates the core requirements of ISO 13485:2016, emphasizing quality throughout the product lifecycle — from design controls to post-market activities. This harmonization reduces regulatory complexity and allows U.S. manufacturers to use one quality management system globally.
Key Similarities Between QMSR and ISO 13485
Both systems require:
- Documented risk management processes.
- Supplier quality control and validation procedures.
- Traceability and design change documentation.
- Management review and CAPA (Corrective and Preventive Action) processes.
Major Differences Manufacturers Must Note
While QMSR aligns with ISO 13485, it retains certain FDA-specific clauses, such as:
- FDA reporting requirements for complaints and adverse events.
- Device history records (DHR) and device master records (DMR).
- Unique Device Identification (UDI) and labeling provisions.
How the QMSR Will Impact Medical Device Quality Systems
The QMSR will require companies to elevate their quality systems with a focus on risk-based thinking, lifecycle management, and digital documentation.
Key areas of change include:
- Expanded design control expectations.
- Real-time data monitoring for CAPA systems.
- Integration of post-market and complaint handling systems.
- Enhanced supplier quality management and verification processes.
Preparing for QMSR Compliance: A Step-by-Step Guide
- Conduct a Gap Analysis: Compare your current QSR system with ISO 13485 and identify nonconformities.
- Revise Procedures and SOPs: Update internal documents, quality manuals, and CAPA procedures.
- Train Teams: Educate staff on QMSR principles and ISO 13485 alignment.
- Leverage Technology: Use digital QMS tools for documentation and traceability.
- Engage with FDA Guidance: Review official updates and attend FDA training sessions.
Common Challenges and How to Overcome Them
Transitioning from the QSR to the new QMSR framework is not a quick or easy process for most manufacturers. It requires a shift in mindset, processes, and documentation. Here are some of the most common challenges — and how to navigate them effectively.
1. Legacy System Upgrades and Documentation Overhauls
Many U.S. manufacturers still rely on outdated documentation systems or paper-based records. Under the QMSR, traceability and documentation integrity are non-negotiable.
Solution: Transition to a digital QMS (eQMS) that automates document control, audit trails, and record retention. This ensures compliance and reduces administrative errors.
2. Managing Dual Compliance During the Transition Period
Until full QMSR implementation in 2026, companies must maintain compliance with both current QSR and ISO 13485requirements.
Solution: Use a dual mapping matrix that aligns QSR clauses with corresponding ISO 13485 elements. This approach minimizes redundancy and prepares the organization for seamless transition.
3. Building a Culture of Continuous Quality Improvement
The FDA’s shift emphasizes quality as a living process, not a checklist.
Solution: Empower teams to take ownership of quality outcomes. Incorporate regular internal audits, feedback loops, and data-driven CAPA initiatives to embed continuous improvement across departments.
Benefits of QMSR Adoption for U.S. Medical Device Companies
The 2026 QMSR transition is not just a compliance requirement — it’s a strategic opportunity. Companies that adapt early will unlock measurable advantages.
1. Global Market Access and Regulatory Streamlining
By aligning with ISO 13485, manufacturers can reduce redundant audits from foreign regulators like the European Medicines Agency (EMA) or Health Canada. This streamlines product approvals and accelerates time-to-market for global launches.
2. Enhanced Risk Management and Product Safety
The QMSR integrates risk-based decision-making into every phase — from design validation to post-market surveillance. This proactive approach significantly reduces adverse events, recalls, and product liability risks.
3. Strengthening Trust with Patients and Healthcare Providers
A robust, ISO-aligned QMS builds credibility. It signals that a manufacturer adheres to the highest global quality standards, strengthening relationships with hospitals, clinicians, and patients who depend on device reliability.
Tools and Technologies Supporting QMSR Implementation
Digital transformation will play a critical role in achieving QMSR compliance efficiently.
Digital QMS Platforms and AI-Driven Compliance Monitoring
Modern platforms like Greenlight Guru, MasterControl, and Qualio enable real-time visibility into quality metrics. Many now integrate AI-based predictive analytics, alerting manufacturers to potential quality deviations before they escalate.

Cloud-Based Documentation and Audit Readiness Tools
Cloud-based systems offer secure, centralized access to all compliance documentation — a major advantage during FDA inspections. Automated workflows ensure revision control and maintain compliance continuity.
Training Resources and FDA Guidance Updates
The FDA has committed to providing training materials, webinars, and updated guidance documents ahead of the 2026 deadline. Leveraging these resources helps organizations stay ahead of regulatory expectations.
???? You can explore official FDA updates at FDA’s QMSR Resource Page.
Expert Insights: What Industry Leaders Are Saying
Perspectives from Quality Managers and Regulatory Experts
Industry leaders view this as a positive evolution. Dr. Karen Thompson, Regulatory Affairs Director at MedTech Insights, notes:
“The QMSR will unify global compliance expectations and cut redundant audits. For U.S. companies, it’s an opportunity to modernize their quality systems for the global stage.”
Predictions for the 2026 Medical Device Landscape
Analysts predict that by 2026, more than 85% of U.S. device manufacturers will adopt hybrid digital quality systems. Those that transition early will experience faster FDA approvals, fewer compliance citations, and stronger international competitiveness.
FAQs: FDA QMSR 2026 and ISO 13485 Compliance
Q1. What is the main goal of the FDA’s QMSR?
The goal is to align U.S. medical device regulations with ISO 13485:2016, reducing redundant audits and improving global consistency in product quality.
Q2. When does QMSR officially take effect?
The final rule will be enforced starting February 2026, replacing the existing QSR framework.
Q3. Will FDA inspections change after QMSR adoption?
Yes. Inspections will focus more on risk management, lifecycle documentation, and quality data integrity aligned with ISO 13485 practices.
Q4. Can ISO 13485 certification guarantee FDA QMSR compliance?
Not entirely. While ISO 13485 alignment covers most requirements, manufacturers must still meet FDA-specific clausessuch as complaint handling, record retention, and labeling requirements.
Q5. What are the penalties for non-compliance?
Failure to comply can result in FDA warning letters, product holds, recalls, or import/export restrictions.
Q6. How should small manufacturers prepare?
Small firms should start early, use cloud-based QMS tools, and seek FDA training webinars to ensure readiness by 2026.
Conclusion: Building a Future-Proof Quality System
The FDA’s 2026 QMSR shift is more than a regulatory update — it’s a transformation of the entire U.S. medical device quality landscape. By aligning with ISO 13485, the FDA is paving the way for safer devices, global harmonization, and data-driven quality systems.
For manufacturers, success will depend on early action, digital modernization, and a strong culture of compliance. Those who invest now will lead the next era of medical device innovation with confidence, agility, and global reach.